Help Transform Recovery1‑3

Jump to
Safety information was collected from 848 OPIOID-DEPENDENT patients across the clinical development program1
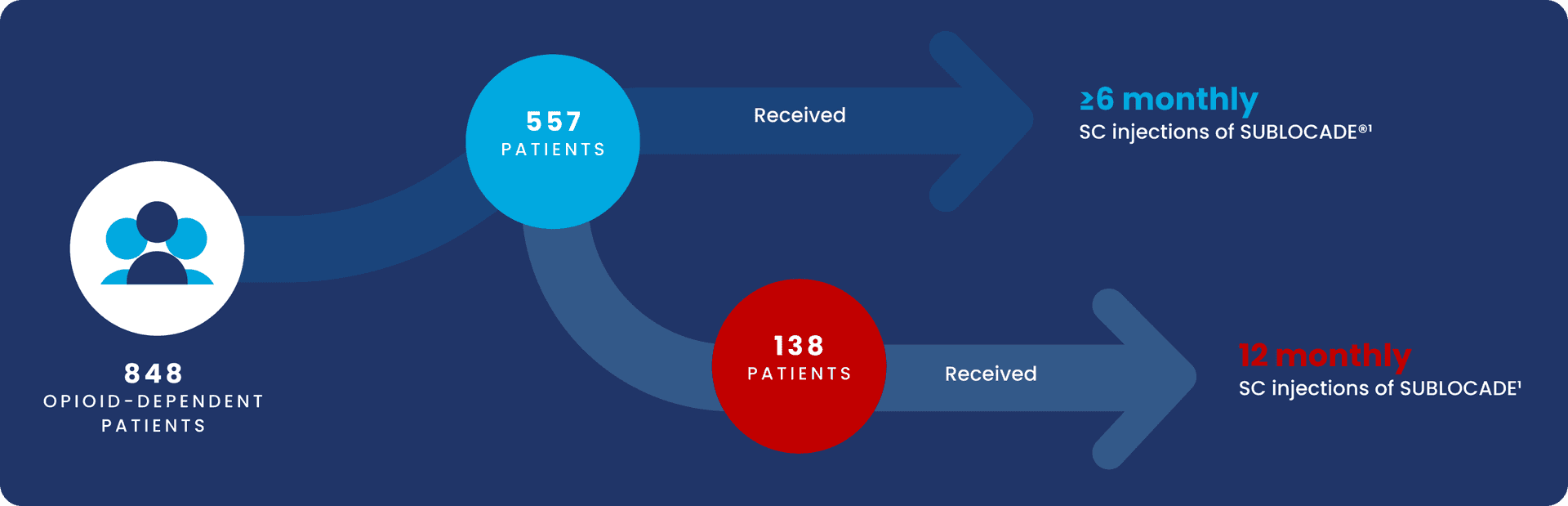
Open‑label 12‑month safety study: overall adverse event profile was similar to the double‑blind trial1,4
- TEAEs leading to SUBLOCADE dose reductions were reported in 7% of participants (47/669).5
- Overall, 67% of participants (449/669) reported any TEAEs.4
- No fatal overdoses were reported.4
Phase 4 open-label rapid induction substudy of 2 injections (N=729, safety population): overall safety profile was generally consistent between rapid initiation and standard induction regimens5
The proportion of patients who experienced an AE associated with opioid withdrawal was reported in 31.4% and 25.1% of patients in the rapid initiation* and standard induction† arm, respectively.1,5
- 3/310 (<1%) of fentanyl-positive patients in the rapid initiation arm who received the first injection of SUBLOCADE had SAEs associated with precipitated withdrawal; there were none in the standard induction arm1,5
*Rapid initiation: 1st SUBLOCADE injection given after a dose of TM BUP (4 mg), for tolerability.1
†Standard induction: 1st SUBLOCADE injection given after ≥7 days of TM BUP.1
AE=adverse event; SAE=serious adverse event; SC=subcutaneous; TEAE=treatment-emergent adverse event.
The systemic safety profile of SUBLOCADE was consistent with the known safety profile of TM BUP1
Adverse Drug Reactions (≥2% of Patients Receiving SUBLOCADE)* in the Phase 3, Double‑Blind Study1
| System Organ Class Preferred Term | SUBLOCADE 300 mg/100 mg n=203 (%) | SUBLOCADE 300 mg/300 mg n=201 (%) | Placebo n=100 (%) |
|---|---|---|---|
| Gastrointestinal disorders | 51 (25.1) | 45 (22.4) | 12 (12) |
| Constipation | 19 (9.4) | 16 (8) | 0 |
| Nausea | 18 (8.9) | 16 (8) | 5 (5) |
| Vomiting | 19 (9.4) | 11 (5.5) | 4 (4) |
| General disorders and administration site conditions | 40 (19.7) | 49 (24.4) | 17 (17) |
| Fatigue | 8 (3.9) | 12 (6) | 3 (3) |
| Investigations† | 21 (10.3) | 19 (9.5) | 2 (2) |
| ALT increased | 2 (1) | 10 (5) | 0 |
| AST increased | 7 (3.4) | 9 (4.5) | 0 |
| CPK increased | 11 (5.4) | 5 (2.5) | 1 (1) |
| GGT increased | 6 (3) | 8 (4) | 1 (1) |
| Nervous system disorders | 35 (17.2) | 25 (12.4) | 7 (7) |
| Headache | 19 (9.4) | 17 (8.5) | 6 (6) |
| Sedation | 7 (3.4) | 3 (1.5) | 0 |
| Dizziness | 5 (2.5) | 3 (1.5) | 2 (2) |
| Somnolence | 10 (4.9) | 4 (2) | 0 |
*SUBLOCADE was given by a healthcare provider in clinical trials.1
†There were no cases of serious liver injury attributed to study drug.1
ALT=alanine aminotransferase; AST=aspartate aminotransferase; CPK=creatine phosphokinase; GGT=gamma-glutamyl transferase; TM BUP=transmucosal buprenorphine.
- Percentages of patients reporting TEAEs in the SUBLOCADE 300 mg/100 mg, SUBLOCADE 300 mg/300 mg, and placebo groups were 76.4%, 66.7%, and 56.0%, respectively5
- TEAEs that led to discontinuation of SUBLOCADE were reported in a similar percentage of patients in the SUBLOCADE 300 mg/100 mg and SUBLOCADE 300 mg/300 mg groups compared with the placebo group (3.0% and 5.0% vs 2.0%, respectively)7
- Dose‑dependent hepatic effects observed in the Phase 3, double‑blind study included the incidence of ALT more than 3 times the upper limit of normal (>3 × ULN) in 5.4%, 12.4%, and 4.0% of the SUBLOCADE 300 mg/100 mg, SUBLOCADE 300 mg/300 mg, and placebo groups, respectively. The incidence of AST >3 × ULN was 7.9%, 11.4%, and 1.0%, respectively1
- There were no cases of serious liver injury attributed to SUBLOCADE1
- Across the development program, 1 subject was withdrawn due to a serious adverse event (abnormal liver function test)5
- There were no deaths related to overdose7
TEAE=treatment-emergent adverse event; ULN=upper limit of normal.
Injection‐site reactions reported for SUBLOCADE1
In the Phase 3 studies, most injection site-related adverse drug reactions were of mild to moderate severity, with 1 report of severe injection‐site pruritus. There were no serious injection site reactions. One reaction, an injection site ulcer, led to study treatment discontinuation1*
Injection Site Adverse Drug Reactions Reported by ≥2% of Patients in the Phase 3, Double‑Blind Study1
| Preferred Term | SUBLOCADE 300 mg/100 mg n=203 (%) | SUBLOCADE 300 mg/300 mg n=201 (%) | Placebo n=100 (%) |
|---|---|---|---|
| Patients with any injection site reactions | 28 (13.8) | 38 (18.9) | 9 (9.0) |
| Injection site pain | 10 (4.9) | 12 (6.0) | 3 (3.0) |
| Injection site pruritus | 13 (6.4) | 19 (9.5) | 4 (4.0) |
| Injection site erythema | 9 (4.4) | 6 (3.0) | 0 |
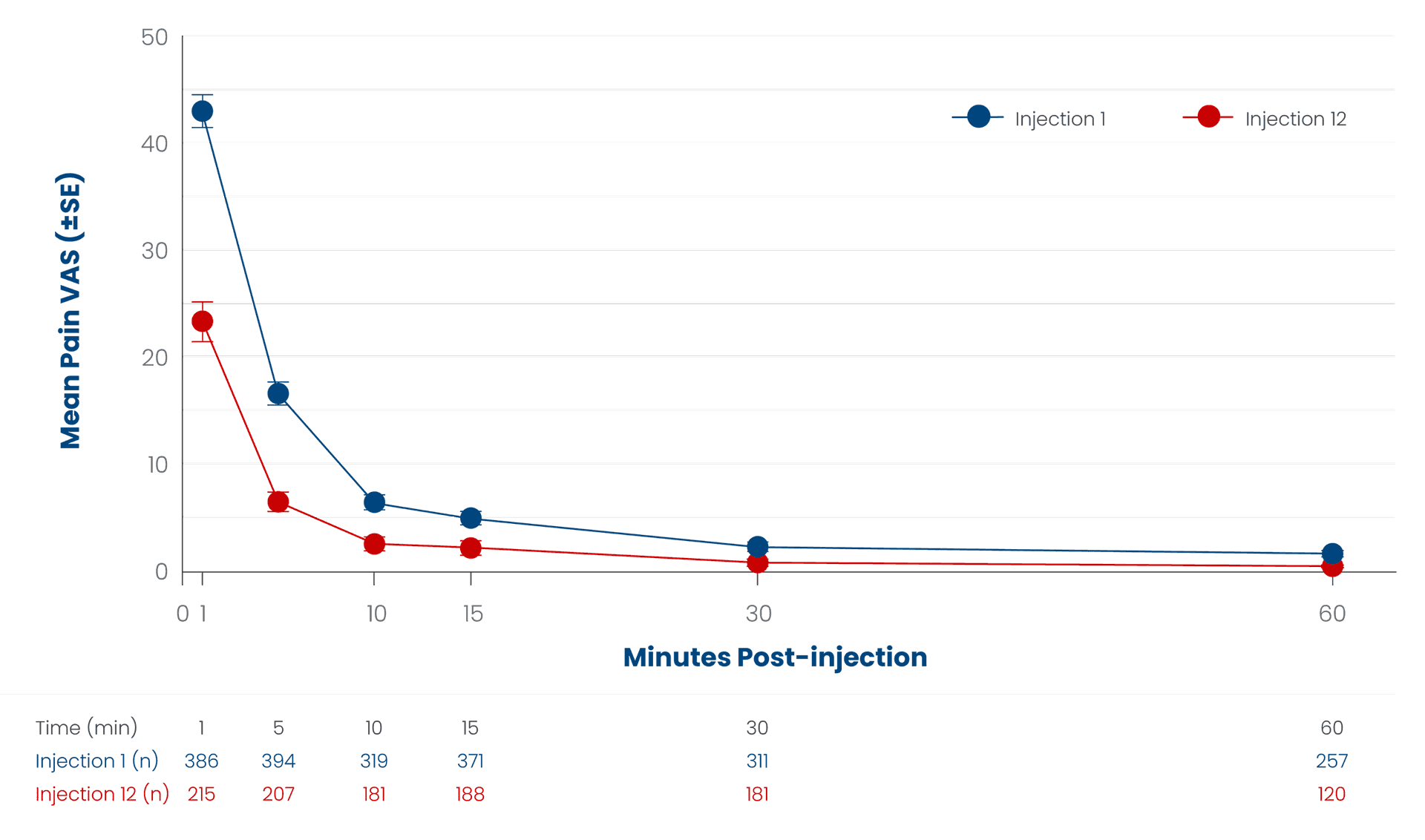
Injection‐site pain was measured in the 12‑month open‑label safety study. Data shown for de novo patients enrolled who received up to 12 SUBLOCADE subcutaneous abdominal injections. Injection‑site pain was measured using the injection‑site pain visual analog scale (VAS), with responses (0‑100 mm) ranging from "no pain" to "pain as bad as it could be". The mean pain scores are shown for Injections 1 and 12.4
SE=standard error.
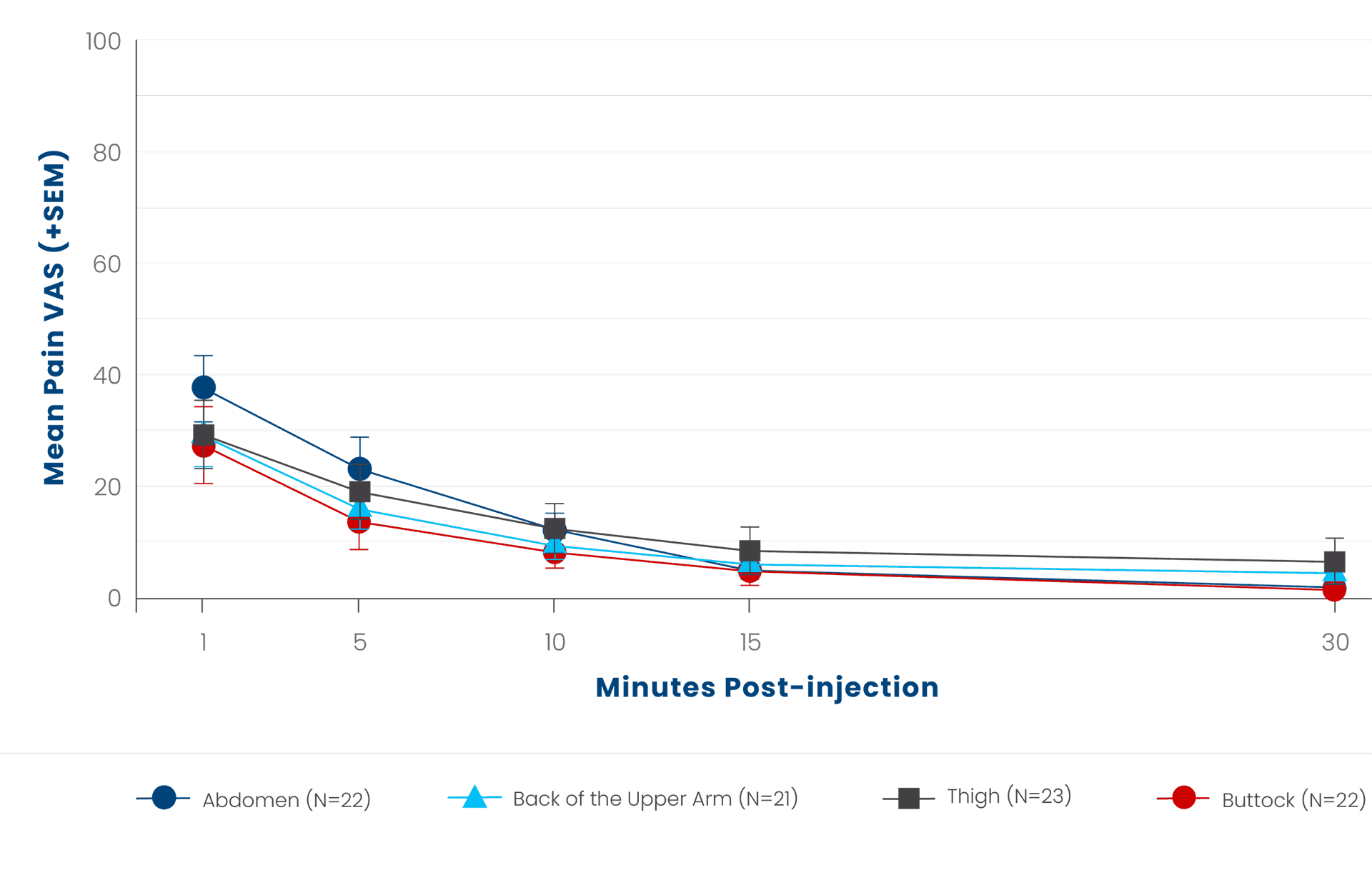
Injection‐site pain was measured in the Phase 4 study that evaluated relative bioavailability (primary objective), safety, and tolerability of a single subcutaneous injection of SUBLOCADE at each of 3 injection‐site locations (back of upper arm, thigh, buttock) compared with the abdomen. Injection‐site pain was measured using a subject-reported visual analog scale (VAS) from 1 to 100 mm, where 0=no pain and 100=maximum pain.5,6
SEM=standard error of the mean.
*All SUBLOCADE injections in the studies were given subcutaneously in the abdomen.5
†Study evaluated the relative bioavailability (primary objective), safety, and tolerability of the alternative sites compared with the abdomen.6
‡Safety results were summarized using descriptive statistics and any differences between alternative injection locations vs the abdomen were estimated with a 95% CI.6
AE=adverse event; CI=confidence interval.
INDICATION
SUBLOCADE is for moderate to severe opioid use disorder in those who have initiated treatment with a dose of transmucosal buprenorphine or are being treated with buprenorphine and should include counseling and psychosocial support.
IMPORTANT SAFETY INFORMATION
WARNING: RISK OF SERIOUS HARM OR DEATH WITH INTRAVENOUS ADMINISTRATION; SUBLOCADE RISK EVALUATION AND MITIGATION STRATEGY
- Serious harm or death could result if administered intravenously. SUBLOCADE forms a solid mass upon contact with body fluids and may cause occlusion, local tissue damage, and thrombo-embolic events, including life threatening pulmonary emboli, if administered intravenously.
- Because of the risk of serious harm or death that could result from intravenous self-administration, SUBLOCADE is only available through a restricted program called the SUBLOCADE REMS Program. Healthcare settings and pharmacies that order and dispense SUBLOCADE must be certified in this program and comply with the REMS requirements.
CONTRAINDICATIONS: SUBLOCADE should not be administered to patients who are hypersensitive to buprenorphine or any component of the delivery system.
WARNINGS AND PRECAUTIONS
Addiction, Abuse, and Misuse: SUBLOCADE contains buprenorphine, a Schedule III controlled substance that can be abused in a manner similar to other opioids. Buprenorphine is sought by people with opioid use disorder and is subject to criminal diversion. Monitor patients for conditions indicative of diversion or progression of opioid dependence and addictive behaviors.
Risk of Life-Threatening Respiratory Depression and Concomitant Use of Benzodiazepines or Other CNS Depressants with Buprenorphine: Buprenorphine has been associated with life-threatening respiratory depression, overdose, and death, particularly when misused by self-injection or with concomitant use of benzodiazepines or other CNS depressants (e.g., alcohol, non‑benzodiazepine sedative/hypnotics, anxiolytics, tranquilizers, muscle relaxants, general anesthetics, antipsychotics, gabapentinoids [gabapentin or pregabalin], and other opioids). Warn patients of the potential danger of self-administration of benzodiazepines, other CNS depressants, opioid analgesics, and alcohol while under treatment with SUBLOCADE. Counsel patients that such medications should not be used concomitantly unless supervised by a healthcare provider.
Use with caution in patients with compromised respiratory function (e.g., chronic obstructive pulmonary disease, cor pulmonale, decreased respiratory reserve, hypoxia, hypercapnia, or pre-existing respiratory depression).
Opioids can cause sleep-related breathing disorders, e.g., central sleep apnea (CSA), sleep-related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. Consider decreasing the opioid using best practices for opioid taper if CSA occurs.
Strongly consider recommending or prescribing an opioid overdose reversal agent (e.g., naloxone, nalmefene) at the time SUBLOCADE is initiated or renewed because patients being treated for opioid use disorder have the potential for relapse, putting them at risk for opioid overdose. Educate patients and caregivers on how to recognize respiratory depression and, if a reversal agent is recommended or prescribed, how to treat an opioid overdose. Emphasize the importance of calling 911 or getting emergency help, even if an opioid overdose reversal agent is administered.
Risk of Serious Injection Site Reactions: The most common injection site reactions are pain, erythema and pruritus with some involving abscess, ulceration, and necrosis. Some cases resulted in surgical depot removal, debridement, antibiotic administration, and SUBLOCADE discontinuation. The likelihood of serious injection site reactions may increase with inadvertent intramuscular or intradermal administration. Carefully review injection technique.
Neonatal Opioid Withdrawal Syndrome: Neonatal opioid withdrawal syndrome (NOWS) is an expected and treatable outcome of prolonged use of opioids during pregnancy. NOWS may be life-threatening if not recognized and treated in the neonate. Newborns should be observed for signs of NOWS and managed accordingly. Advise pregnant women receiving opioid addiction treatment with SUBLOCADE of the risk of neonatal opioid withdrawal syndrome.
Adrenal Insufficiency: Adrenal insufficiency has been reported with opioid use. If adrenal insufficiency is diagnosed, treat with physiologic replacement doses of corticosteroids. Wean the patient off the opioid.
Discontinuation of SUBLOCADE Treatment: Due to the long-acting nature of SUBLOCADE, if treatment is discontinued, monitor patients for several months for withdrawal and treat appropriately.
Inform patients that they may have detectable levels of buprenorphine for a prolonged period of time after treatment with SUBLOCADE. Considerations of drug-drug interactions, buprenorphine effects, and analgesia may continue to be relevant for several months after the last injection.
Risk of Hepatitis, Hepatic Events: Because cases of cytolytic hepatitis and hepatitis with jaundice have been observed in individuals receiving buprenorphine, monitor liver function tests prior to treatment and monthly during treatment.
Hypersensitivity Reactions: Hypersensitivity to buprenorphine-containing products have been reported most commonly as rashes, hives, and pruritus. Some cases of bronchospasm, angioneurotic edema, and anaphylactic shock have also been reported.
Precipitation of Opioid Withdrawal in Patients Dependent on Full Agonist Opioids: Buprenorphine may precipitate opioid withdrawal signs and symptoms in persons who are currently physically dependent on full opioid agonists if administered before the effects have subsided, at least 6 hours for short-acting opioids and 24 hours for long-acting opioids. Verify that patients have tolerated transmucosal buprenorphine before administering the first injection of SUBLOCADE.
Risks Associated With Treatment of Emergent Acute Pain: When patients need acute pain management, or may require anesthesia, treat patients receiving SUBLOCADE currently or within the last 6 months with a non-opioid analgesic whenever possible. If opioid therapy is required, patients may be treated with a high-affinity full opioid analgesic under the supervision of a physician, with particular attention to respiratory function, as higher doses may be required for analgesic effect and therefore, a higher potential for toxicity exists with opioid administration.
Advise patients of the importance of instructing their family members, in the event of emergency, to inform the treating healthcare provider or emergency room staff that the patient is physically dependent on an opioid and that the patient is being treated with SUBLOCADE.
Use in Opioid Naïve Patients: Because death has been reported for opioid naïve individuals who received buprenorphine sublingual tablet, SUBLOCADE is not appropriate for use in opioid naïve patients.
Use in Patients With Impaired Hepatic Function: Because buprenorphine levels cannot be rapidly decreased, SUBLOCADE is not recommended for patients with pre-existing moderate to severe hepatic impairment. Patients who develop moderate to severe hepatic impairment while being treated with SUBLOCADE should be monitored for several months for signs and symptoms of toxicity or overdose caused by increased levels of buprenorphine.
QTc Prolongation: QT studies with buprenorphine products have demonstrated QT prolongation ≤ 15 msec. Buprenorphine is unlikely to be pro-arrhythmic when used alone in patients without risk factors. The risk of combining buprenorphine with other QT-prolonging agents is not known. Consider these observations when prescribing SUBLOCADE to patients with risk factors such as hypokalemia, bradycardia, recent conversion from atrial fibrillation, congestive heart failure, digitalis therapy, baseline QT prolongation, subclinical long-QT syndrome, or severe hypomagnesemia.
Impairment of Ability to Drive or Operate Machinery: SUBLOCADE may impair the mental or physical abilities required for the performance of potentially dangerous tasks such as driving a car or operating machinery. Caution patients about driving or operating hazardous machinery until they are reasonably certain that SUBLOCADE does not adversely affect their ability to engage in such activities.
Orthostatic Hypotension: Buprenorphine may produce orthostatic hypotension.
Elevation of Cerebrospinal Fluid Pressure: Buprenorphine may elevate cerebrospinal fluid pressure and should be used with caution in patients with head injury, intracranial lesions, and other circumstances when cerebrospinal pressure may be increased. Buprenorphine can produce miosis and changes in the level of consciousness that may interfere with patient evaluation.
Elevation of Intracholedochal Pressure: Buprenorphine has been shown to increase intracholedochal pressure, as do other opioids, and thus should be administered with caution to patients with dysfunction of the biliary tract.
Effects in Acute Abdominal Conditions: Buprenorphine may obscure the diagnosis or clinical course of patients with acute abdominal conditions.
Unintentional Pediatric Exposure: Buprenorphine can cause severe, possibly fatal, respiratory depression in children who are accidentally exposed to it.
ADVERSE REACTIONS: Adverse reactions commonly associated with SUBLOCADE (≥5% of subjects) during clinical trials were constipation, headache, nausea, vomiting, increased hepatic enzymes, fatigue, and injection site pain and pruritus. This is not a complete list of potential adverse events. Please see the full Prescribing Information for a complete list.
DRUG INTERACTIONS
CYP3A4 Inhibitors and Inducers: Monitor patients starting or ending CYP3A4 inhibitors or inducers for potential over- or under-dosing.
Serotonergic Drugs: If concomitant use with serotonergic drugs is warranted, monitor for serotonin syndrome, particularly during treatment initiation, and during dose adjustment of the serotonergic drug.
Consult the full Prescribing Information for SUBLOCADE for more information on potentially significant drug interactions.
USE IN SPECIFIC POPULATIONS
Pregnancy: Opioid-dependent women on buprenorphine maintenance therapy may require additional analgesia during labor.
Lactation: Buprenorphine passes into the mother's milk. Advise breastfeeding women to monitor the infant for increased drowsiness and breathing difficulties.
Fertility: Chronic use of opioids may cause reduced fertility. It is not known whether these effects on fertility are reversible.
Geriatric Patients: Monitor geriatric patients receiving SUBLOCADE for sedation or respiratory depression.
To report a pregnancy or side effects associated with taking SUBLOCADE or any safety related information, product complaint, request for medical information, or product query, please contact PatientSafetyNA@indivior.com or 1-877-782-6966.
See full Prescribing Information, including Boxed Warning, and Medication Guide. For REMS information visit www.sublocadeREMS.com.
P-BAG-US-01758 April 2025